|
12/23/2023 0 Comments Graphical analysis  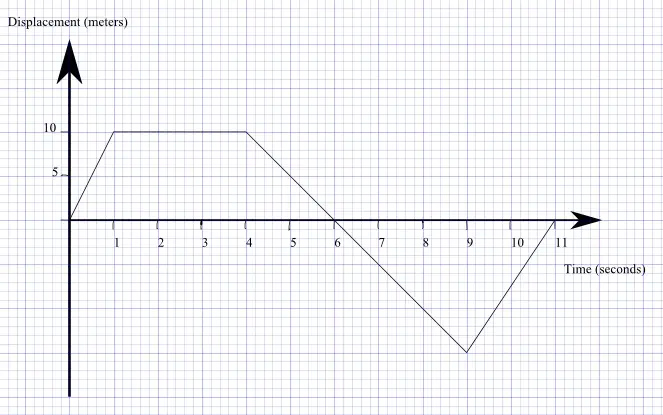 
Let us construct a graph from data that one might collect in an experiment. One contains independent variable values, usually in numerical order the other contains the corresponding dependent variable values.įigure 4. Before plotting the data, it is often convenient to list the data in two columns. The variable plotted along the x axis is called the independent variable the variable plotted along the y axis is called the dependent variable. The point is located 21 units to the right of the y axis (that is, 21 units along the x axis) and 15.0 units above the x axis (that is, 15.0 units along the y axis). Figure 4 shows how this is done with the point for the element scandium: Z = 21 and V = 15.0 mL/mol. Each value of Z is plotted along the horizontal axis, also called the x axis or abscissa, and the corresponding value of V is plotted along the vertical axis, also called the y axis or ordinate. Each point on this graph represents an ordered pair of data: for each value of Z there is a corresponding value of V. 1, the variables are atomic number (Z) and molar volume (V). An xy graph is used to display the relationship between two variables. The remainder of this exercise will concentrate on xy graphs. Pie charts are particularly well suited to display of the partition of a whole among a small number of parts. United States energy consumption by sector in 1999.įigure 3 displays US energy consumption in various energy sectors as a percentage of total consumption. Note how just glancing at this simple chart clearly divides the samples into two sets with slightly but definitely different values.įigure 3. Mass of nitrogen, derived from various sources, contained in a standard glass globe.įigure 2 shows the mass of nitrogen gas contained in a certain glass globe the labels on the vertical axis denote various methods for preparing the sample. Bar and column graphs are frequently used to display values of a single quantity measured for different samples when the samples differ in kind rather than in number.įigure 2. 2 (and the closely related column graph, which uses vertical rather than horizontal bars) and the pie chart, shown in Fig. Other common graph formats include the bar graph, shown in Fig. This is the most common format for plotting scientific data, as it displays the relationship between two variables. Note that the peaks in this graph all belong to alkali metals.įigure 1 is an example of an xy graph, sometimes also called a scatter plot or a line graph. Graph of molar volume (mL per mole of atoms) of elemental solids vs. Note that the peaks are elements that fall in the same column in the modern table.įigure 1. The plot below shows how one property of elements, the molar volume, rises and falls with atomic number. In the case of chemical periodicity-the repeated rise and fall of chemical properties with atomic number that forms the basis of the periodic table-a graphical representation shows relationships that might not be apparent otherwise. 
For example, questions about patterns in the data and whether one variable changes when another changes are often easier to see in a graph than in a table. This exercise will give students practice in constructing graphs that clearly convey experimental data.īackground: Graphs and quantitative data Communicating and even seeing information about numerical data can be enhanced by graphical display. They can also be used to derive quantitative relationships between variables. ObjectGraphs can be used to display large amounts of numerical data and to portray the relationships between numerical values of different variables. Graphical Analysis and Display of Data Graphical Analysis and Display of Data Ĭhemical Principles Laboratoryby Carmen Giunta, Le Moyne College 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
